The World of Amino acids and Proteins #3
Hello, dear readers. Thanks for all your comments, supports and upvotes. I do appreciate y'all. Today, on my series: The World of Amino acids and Proteins, I will be starting with the structure of protein and later discuss about enzymes being catalysts.
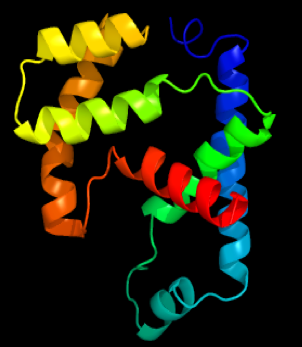
PROTEIN STRUCTURE
Proteins are very large molecules, with values of Mr that range from 5000 to several million. When proteins are formed inside a cell, they are too large to pass through the cell membrane and so stay trapped inside. That is, unless tissues are damaged. For example, certain enzymes (which we have seen are proteins) can be found outside cells after a heart attack. Normally, these enzymes would be confined inside the cells of the heart, but during a heart attack some of the cells rupture, which allows these enzymes to escape. The more major the heart attack, the more cells split open and the higher the concentration of the enzymes found in the blood. The presence of these enzymes in the blood is used by doctors to diagnose the seriousness of a heart attack. In the same way, certain proteins in urine indicate that a kidney has been damaged.
PRIMARY STRUCTURE OF PROTEINS
Although only 20 amino acids, usually, make up our proteins, the number of ways in which they are combined is vast. Take, for example, lysozyme, an enzyme found in tears, saliva, nasal mucous and milk, which destroys the cell walls of bacteria by breaking certain bonds through hydrolysis. It has 129 amino acid residues and uses all 20 amino acids. The number of different arrangements of the 20 amino acids in a protein chain of this length is 20129 (7.0 × 10167), which is estimated to be more than twice the number of atoms in our galaxy.
The sequence of the amino acids in a protein is called its primary structure and this forms the backbone of the protein.
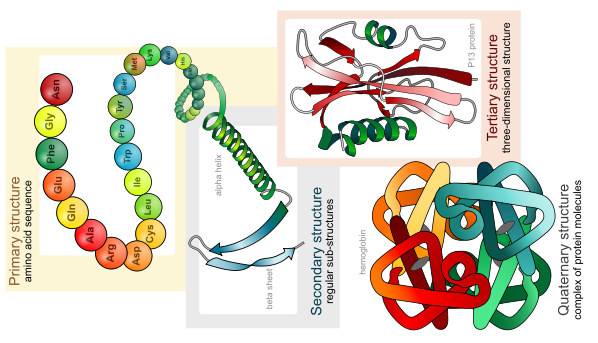
Main protein structures levels. LadyofHats, public domain
HYDROLYSIS OF PEPTIDE BONDS
To find the sequence of amino acids in a protein, the peptide links are hydrolyzed (split by the action of water). This is done by boiling the protein with an acidic or alkaline solution, or by using certain enzymes.
The protein chain may be hydrolyzed completely, to leave a mixture of individual amino acids that can be separated using chromatography. This gives information as to how many different amino acids are present.
If different hydrolyzing agents are used, the protein chain is split in different places. Some of the amino acid sequences overlap, which enables the overall sequence of amino acids to be worked out.
PRIMARY STRUCTURE OF INSULIN
Insulin was the first protein to have its amino acid sequence (primary structure) worked out. This was done by the English biochemist Frederick Sanger, who began in 1944 and spent ten years on the task. One of the techniques he used was to completely hydrolyze the insulin into its component amino acids by heating a mixture of 6 mol dm-3 hydrochloric acid with insulin in a sealed tube for 24 hours.
Sanger then separated the amino acids, using paper chromatography. Instead of using just one solvent too separate the amino acids, he used two, one after the other, in a technique called two-way chromatography. A chromatogram is made using the first solvent and is left to dry. Then it is turned on its side and the second solvent is applied.
For his painstaking research, Sanger received the Nobel Prize for Chemistry in 1958. In 1980, he was awarded a second Nobel Prize jointly with two American scientists for work on DNA, and so he became only the third person in history to win two Nobel prizes in scientific disciplines.

SECONDARY STRUCTURE OF PROTEINS AND HYDROGEN BONDING
The shape of a protein molecule is what makes it able to perform its enzyme function.
Linus Pauling, the eminent American scientist, first turned his attention to protein structure in the 1930s. After 15 years, he and another American, Robert Corey, proposed that the primary structure of proteins could have one of two orderly arrangements, both held together by hydrogen bonds.
The first arrangement is a regular coiling of part of the polypeptide chain, called an α-helix. The second arrangement is a folding of the polypeptide chain to make sheets, called a β-pleated sheet. These are known as the secondary structures of a protein. Both were subsequently discovered through X-ray crystallography.
The hydrogen bonding that holds a secondary structure together is between the N–H of one peptide link and the C=O of another. Since this is within the molecule, it is known as intramolecular hydrogen bonding.
The α-helix is a right-handed spiral and hydrogen bonds form between every fourth amino acid residue. It is found extensively in wool fibres and allows wool to stretch. When it is pulled, the α-helix elongates, which breaks the hydrogen bonds. When the α-helix is released, the hydrogen bonds re-form as the α-helix returns to its usual shape.
The β-pleated sheet occurs when the amino-acid backbone of the primary structure folds to give sections of parallel chains of amino acids, which again form hydrogen bonds through their peptide links.
Although the β-pleated sheet is fairly flexible, it cannot be stretched, as the chains of amino acids are already extended. Silk, for example, is composed of the protein silk fibroin. This is almost entirely β-pleated sheet, which is the reason why silk cannot be stretched like wool.
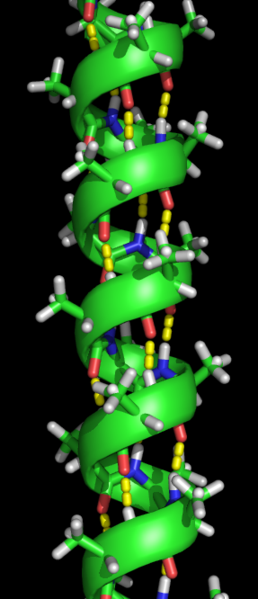
An α-helix with hydrogen bonds (yellow dots). en:User:Bikadi, CC BY-SA 3.0
TERTIARY STRUCTURE OF PROTEINS
The overall three-dimensional shape of a protein is the result of yet more folding and bending to give what looks like a random structure, but most definitely is not. This is the tertiary structure and is specific to a particular polypeptide chain. It is this shape that determines how each protein will function.
Proteins can be divided into two sorts: globular proteins and fibrous proteins.
Globular proteins, as their name implies, are roughly spherical (e.g. myoglobin). Our enzymes and protein hormones are globular proteins, and their polypeptide chains are folded extensively to give a very compact structure. These proteins are usually soluble because they are arranged in a way that leaves the hydrophilic R-groups of individual amino acid residues on the outside of their structures.
Fibrous proteins have their polypeptide chains arranged in bundles to form fibres. They have a structural function. Collagen is a fibrous protein in tendons and muscles that consists of intertwining polypeptide chains.
The α-keratin of hair, wool and claws is a fibrous protein in which the α-helix is twisted so that several strands can intertwine. The silk fibroin protein is also fibrous, only this time β-pleated sheets become bound together.
The tertiary structure of a protein is held together by four different types of interaction between the various R Side chains of the different amino acids.
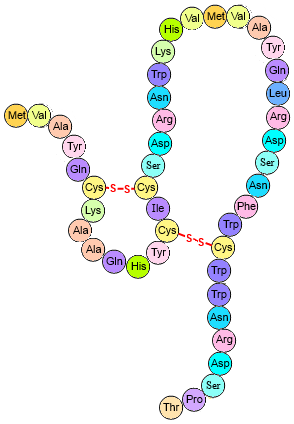
Simple polypeptide showing disulphide bridges. CKRobinson - Own work, CC BY-SA 4.0
It makes your hair curl!
Our hair is made up of strands of the protein α-keratin. Its shape is maintained by hydrogen bonds, ionic linkages and disulphide bridges between the keratin strands. The very act of washing our hair disrupts these forces because water molecules can get in between the strands and affect all three interactions – in particular, the hydrogen bonds.
As hair dries, the water molecules leave the keratin and the original forces reassert themselves. So, if we do not immediately comb and set our hair in the shape we want, hydrogen bonds may re-form to curl our hair in a way we don’t want.
ENZYMES AS CATALYSTS
An enzyme is a bio-molecule catalyst. Almost all enzymes are proteins, and most are found inside cells. Like all catalysts, enzymes alter the energy of activation of a reaction by providing an alternative route for reactants to interact without becoming permanently involved in the reaction. Enzymes work by having a three-dimensional active site that is part of the tertiary structure of the enzyme. The molecule that an enzyme catalyses is called the substrate. The substrate molecule forms intermolecular bonds, such as hydrogen bonds, that bind it to the active site. This reduces the activation energy required for the particular reaction. When the product molecules are formed they have weaker intermolecular forces binding them to the active site, and are able to leave easily to be replaced by more substrate molecules.
Enzymes differ from other catalysts in their amazingly high activity, being between a million and 20 million times more efficient than inorganic catalysts. Sometimes, each enzyme molecule can catalyze the reaction of thousands of molecules per second. An example is the enzyme catalase, which catalyses the decomposition of hydrogen peroxide:
2H2O2(l) → 2H2O (l) + O2 (g)
One molecule of catalase catalyses about 50,000 molecules of H2O2, per second at 0°C, whereas manganese(IV) oxide, an inorganic catalyst for this reaction, works very much less efficiently at 0°C.
Enzymes can also be very specific in the reactions they catalyze. For example, out of the vast number of chemicals in the body, an enzyme may catalyze just one particular reaction of one chemical.
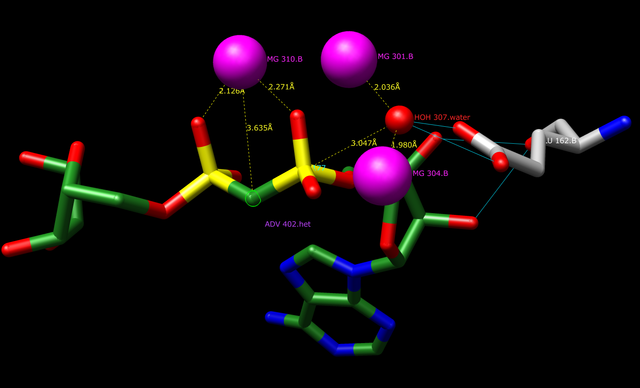
ADP Ribose Diphosphatase Mechanism (Glutamate 162 as catalyst). Darhink (talk), Public Domain
The three-dimensional shape of an enzyme is critical to its catalytic ability. This shape can be denatured (disrupted) by changes of temperature, by altering the pH of the environment of the enzyme or by exposing it to heavy metal ions, such as mercury(II) ions (Hg2+) and lead(II) ions (Pb2+). Enzymes have an optimum temperature and pH at which they work best. Their catalytic activity decreases markedly outside a narrow range, which is why it is dangerous to have a very high temperature for too long during an illness. Most enzymes work efficiently in the range of 25-40 °C, and their shape is irreversibly changed at about 50-60 °C. The optimum pH for most of our Body enzymes is about pH 7, but pepsin, a digestive enzyme, has an optimum pH of 2, with its maximum activity in the acid conditions of the stomach.
It is also possible for molecules, which also fit into the active site and form inter-molecular bonds to it, to inhibit the enzyme’s catalytic ability. As they do not react, the inhibitor molecules stay in the active sites and block them to other substrate molecules.
IMMOBILIZING ENZYMES
At the start of this article, you read about the advantages of using enzymes in industry. Unfortunately, if an enzyme is simply mixed with the reactants to be catalyzed, it is difficult to separate at the end of the reaction and is usually destroyed. However, if the enzyme is attached to an inert surface across which the reactants pass, the enzyme functions as a heterogeneous catalyst and can be used several times over.

Various surfaces can hold the enzyme – from polystyrene to glass beads – and immobilized enzymes are used increasingly in food and chemical manufacture. They are also important in medical diagnosis: clinical tests for glucose and cholesterol rely on immobilized enzymes that react with these compounds.
Thanks for reading.
REFERENCES
https://www.particlesciences.com/news/technical-briefs/2009/protein-structure.html
https://en.wikipedia.org/wiki/Protein_structure
https://www.rsc.org/Education/Teachers/Resources/cfb/ppt/06peptidehydrolysis.pps
https://www.biotopics.co.uk/as/insulinproteinstructure.html
http://www.vivo.colostate.edu/hbooks/pathphys/endocrine/pancreas/insulin_struct.html
https://www.chemguide.co.uk/organicprops/aminoacids/proteinstruct.html
https://en.wikipedia.org/wiki/Protein_secondary_structure
https://en.wikipedia.org/wiki/Protein_tertiary_structure
https://www.ouidad.com/blog/curly-hair-routine-101/hair-protein-products-for-curly-hair
https://www.eolss.net/Sample-Chapters/C03/E6-54-02.pdf
https://en.wikipedia.org/wiki/Enzyme_catalysis
https://www.cliffsnotes.com/study-guides/biology/biochemistry-i/enzymes/enzymes-are-catalysts
https://www.sciencedirect.com/topics/engineering/enzyme-immobilization
https://en.wikipedia.org/wiki/Immobilized_enzyme
Thanks for continuing this!
Regards
Chapper
It's a pleasure. Thanks for the support, Mr. Chapper.
This post has been voted on by the SteemSTEM curation team and voting trail. It is elligible for support from @curie and @utopian-io.
If you appreciate the work we are doing, then consider supporting our witness stem.witness. Additional witness support to the curie witness and utopian-io witness would be appreciated as well.
For additional information please join us on the SteemSTEM discord and to get to know the rest of the community!
Thanks for having added @steemstem as a beneficiary to your post. This granted you a stronger support from SteemSTEM.
Thanks for having used the steemstem.io app. You got a stronger support!
Hi @empressteemah!
Your post was upvoted by Utopian.io in cooperation with @steemstem - supporting knowledge, innovation and technological advancement on the Steem Blockchain.
Contribute to Open Source with utopian.io
Learn how to contribute on our website and join the new open source economy.
Want to chat? Join the Utopian Community on Discord https://discord.gg/h52nFrV
A nice series! Thanks for sharing it here!
It's a pleasure. Thanks for coming by.